Entropy & Second Law of Thermodynamics: Nuts & Bolts
Entropy & Second Law of Thermodynamics: Nuts & Bolts
3. Nuts & Bolts
Algorithm
Expand
Multiple Representations
| Multiple Representations is the idea that a physical phenomena can be explored in many different ways. For example, there is the physical representation which models the system with figures and diagrams, such as a free body diagram. There is also the mathematical representation which uses the equation(s) governing the physics of the system. All of the representations can be used together to help us understand and quantify the physical phenomena. |
Observe the different types of representations for this section below:
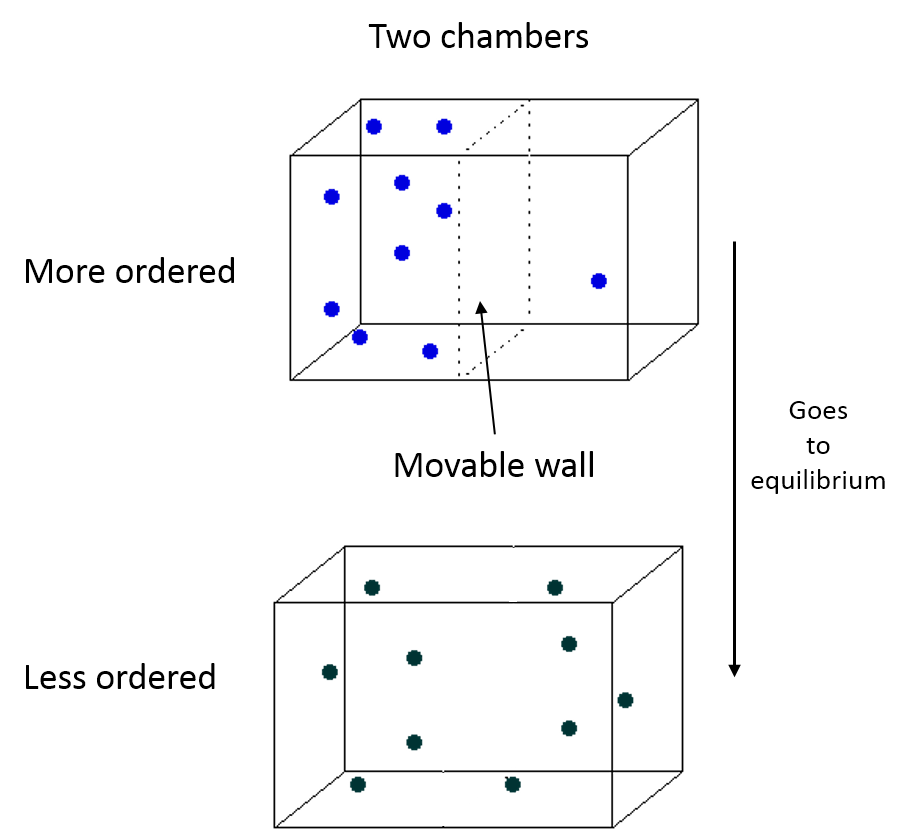
Physical Representation
We have a system with a moveable wall. Once the wall is removed, the particles move toward a new equilirium.
Mathematical Representation
$*Q = \lim_{\Delta S \to 0} \sum T \Delta S $
*Actual equation not important.What is important is that conceptually heat is proportional to the change in entropy.
Conceptually, heat (Q) is from $\Delta S$
Graphical Representation
.
Descriptive Representation
Entropy drives isolated systems, such as the boxes of particles shown in the Physical Representation, to increased disorder. Where more disorder has larger micro multiplicity and a larger multiplicity means more microscopic configurations with the same macroscopic observables such temperature (T), pressure (P), $E_{th}$, etc...
Experimental Representation
We can design an experiment by taking a water filled ballon and popping it. Initially, the water in the balloon is contained and relatively uniform. However, as soon as we pop the balloon the water molecules become scattered and begin to spread out to form a new equilibrium.
Example Problems