Heat: Tips & Tricks
Heat: Tips & Tricks
Problem Solving Guide
Algorithm
General problems
Since heat is a mechanism for transferring thermal energy between systems, expect some heat problems to really be problems about changes in $E_{th}$ according to the First Law. In the previous section you were typically given 2 of the variables out of $Q$, $W$, and $\Delta E$ and asked to calculate the other. In this section you will mostly be using the following two equations:
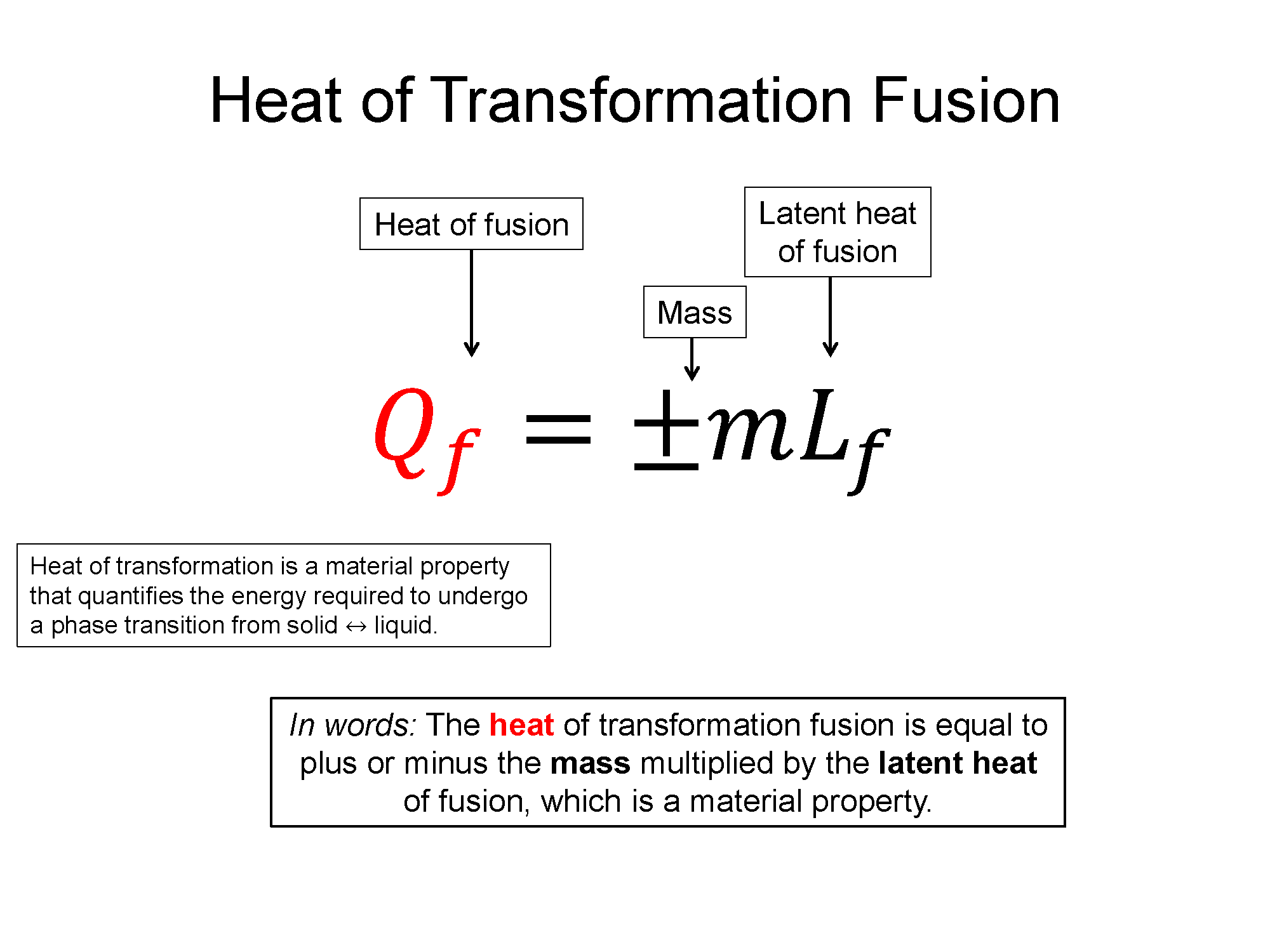
$Q_f = \pm mL_{f,v}$
or
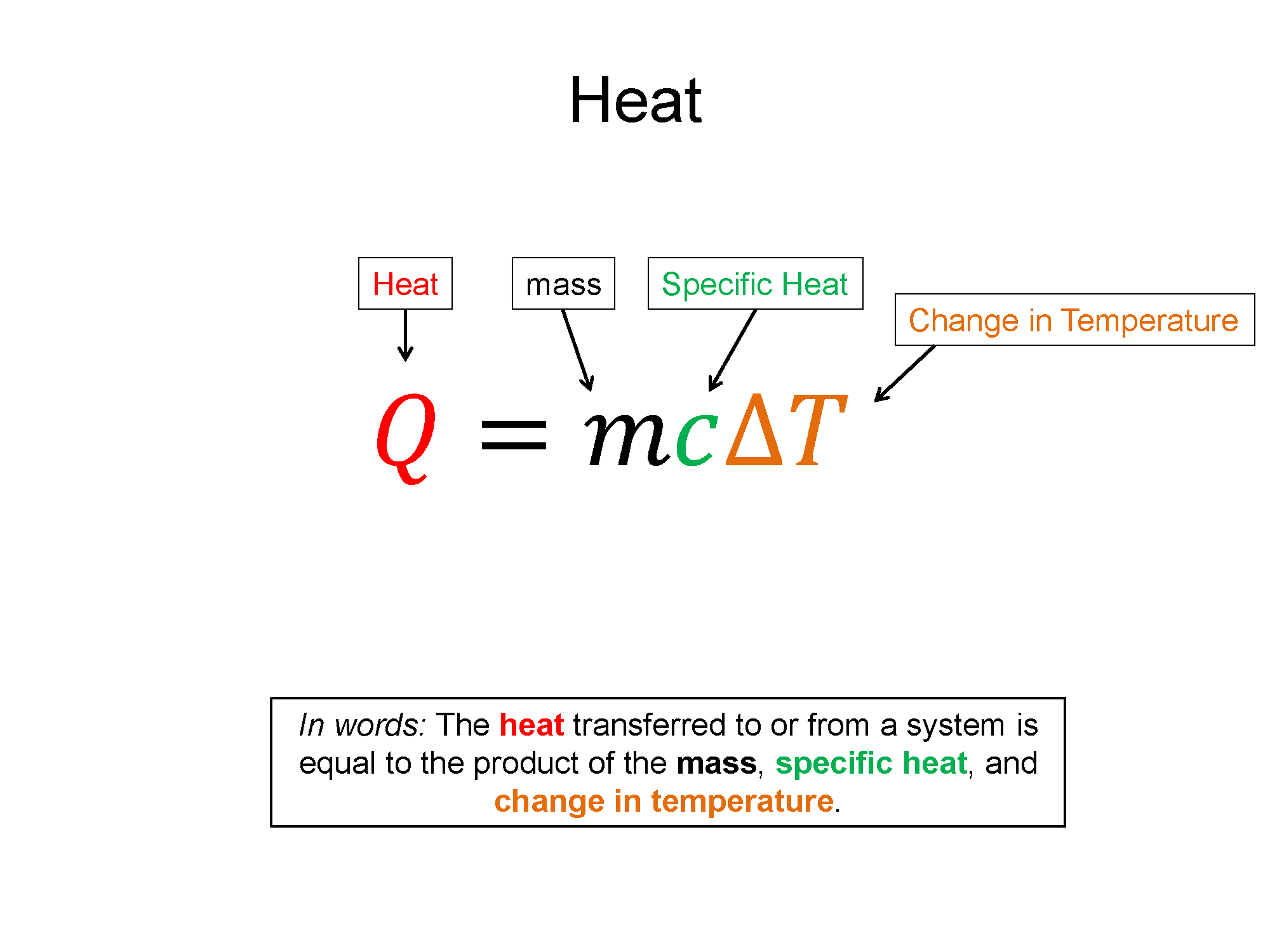
$Q=mc\Delta T$
In general, you will be given a few variables and asked to solve for another - such as being given a mass, heat capacity, and change in temperature of an object, and asked how much thermal energy was transferred into our out of the object as heat. Or, you may be asked, for a given type of object, how much its temperature would change when in contact with a thermal reservoir that transfers some $Q_H$ amount of heat. For most of these problems, read the prompt carefully and take an inventory of your knowns and unknowns and the rest should be straightforward. Being able to do this comfortably will help enormously with calorimetry problems.
Calorimetry
Remember that calorimetry specifically refers to isolated systems with constant volume, so both the change in thermal energy of the system and work done on or by the system is zero ($ \Delta E = W = 0$).
1. For all calorimetry problems, begin by summing the heat flow in into and out of the system:
$\Sigma Q = Q_1 + Q_2 + Q_3 + ... = 0$
2. After summing the sources of heat flow, read the problem statement again and verify that you haven't missed any sources (this is a common mistake students make - misreading a problem and missing that there is a phase change, for example).
3. Re-write the sum in terms of definitions. For each $Q$, plug in its $mc\Delta T$. If phase changes are present and thus you have heats of transformation present, replace $Q$ with $mL_v$ or $mL_f$ with $v$ for vaporization, $f$ for fusion.
For example, pretend you put a hot rock in a container of liquid water and you observe steam. The equation to describe this is
$\Sigma Q = Q_{rock} + Q_{water,l} \pm Q_{v,water} + Q_{water,g}$
which is re-written as
$\Sigma Q = m_{r}c_{r}\Delta T_{r} + m_{w,l}c_{w,l} \Delta T_{w,l} + m_{v,w} L_{v, w} + m_{w,g}c_{w,g}\Delta T_{w,g}$
*The $\pm$ in front of the heat of vaporization term in the first equation was replaced by a $+$ sign in the second equation because we know the transformation was from liquid to vapor, and that is the sign convention for such a phase transition.
4. This equation should include all of the information you need to solve the problem. Note that since the systems are in thermal contact, they will come to thermal equilibrium and thus have the same final temperature $T_f$, but not the same $\Delta T$.
Often the point of a calorimetry problem is to find a final temperature, although sometimes the final temperature is given and you are asked for the heat exchange between particular sub-systems (such as the amount of thermal energy required to produce as much steam as was observed). Remember that the final temperature should be somewhere between the initial temperatures of the sub-systems (liquid water and hot rock); if it is somewhere outside, you probably made a calculation mistake or have a sign error somewhere (check that your $\Delta T = T_f - T_i$ have the correct signs).
Misconceptions & Mistakes
- Heat is a mechanism by which thermal energy is transferred between systems. A system does not "contain" heat - it is not a state variable like P, V, T, N (and S).
- Note that just because a phase change is taking place doesn't mean $W=0$; you can change liquid water to steam by changing the pressure of its container.
Pro Tips
- Practice practice practice. This section looks like a giant hair ball at first (especially calorimetry questions), but ends up being straightforward if you are careful and take your time. You will have lots of variables to keep track of (and lots of them are $Q_{something}$), so the more you practice keeping track of them, the easier your life will be.
Multiple Representations
Multiple Representations is the concept that a physical phenomena can be expressed in different ways.
Physical
Heat transfer occurs from the flame to the water in the cup. It takes energy through heat transfer to change the temperature of the water in the cup. As we add heat to the system the energy increases with respect to time.
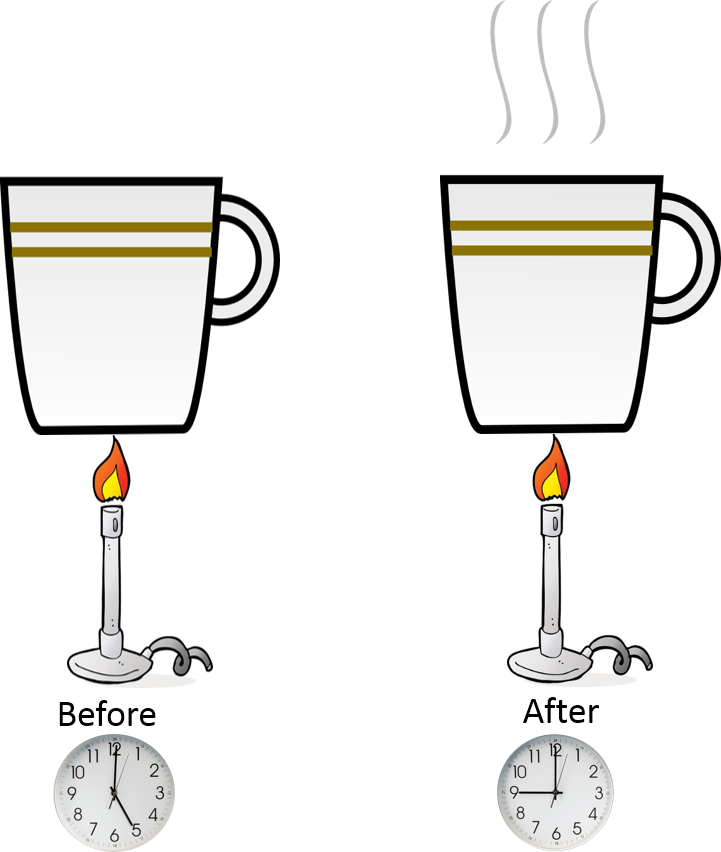
Mathematical
$Q = mc\Delta t$, where c is specific heat
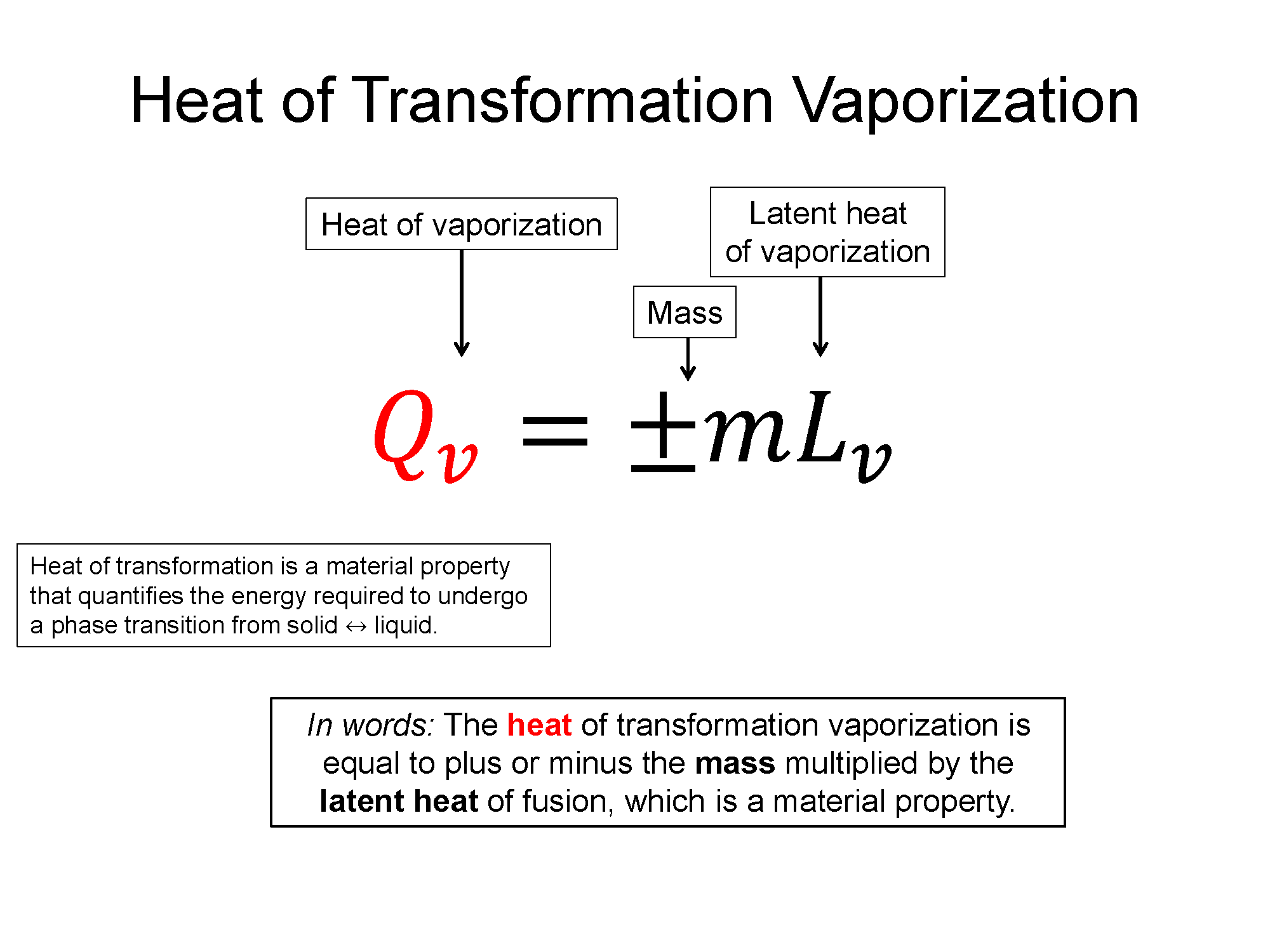
$Q = \pm mL_{f,v}$, where L is latent heat
Calorimetry if all the transfer occurs in an isolated system, then we have the following:
$\Delta Q_{tot} = 0$ then $\sum Q = 0 \implies Q_1 + Q_2+Q_3 ... Q_n =0$

Graphical
The graph describes the general phase transitions for a material. Notice the transition phases from solid to liquid and liquid to gas are constant temperature. Different materials have different temperature values where the phase transition occurs.
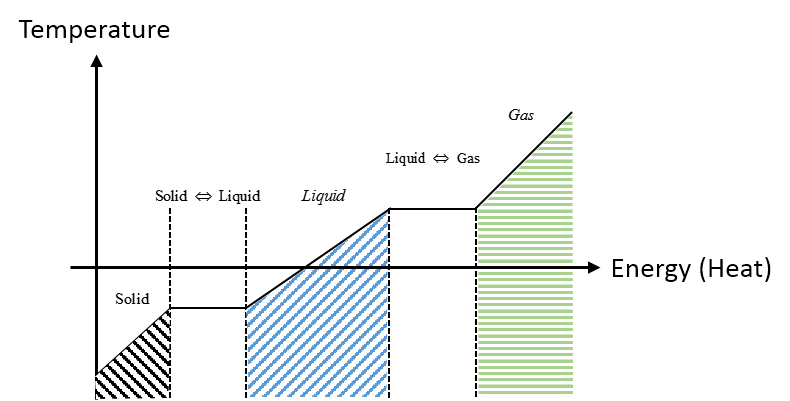
Descriptive
Referring to the Graphical Representation that describes the phase transitions of a material. During the initial solid phase the temperature increases and we add energy to the system until we reach the first phase change where we have reach the temperature required for the material to undergo the first phase transition. The next phase is the liquid to solid transition, where the material requires more added energy to the system to raise the temperature and complete the last phase transition.
Experimental
We can begin with an ice cube, a closed lid cup, a thermometer, and a bunsen burner. We place the ice cube inside the cup and insert the thermometer and close the lid around the thermometers. Place the the cup over the bunsen burner and ignite the flame. Record the temperature and observe the state of the matter. We can then use this information to plot a phase change diagram as depicted in the Graphical representation and observe the temperature as it relates to the energy required for the ice to undergo transitions from ice to water to gas.