Thermodynamic Cycles: Tips & Tricks
Thermodynamic Cycles: Tips & Tricks
Problem Solving Guide
Algorithm
Begin filling in the table with "non-calculation" values - values you can read right off the plot.
Use proportional reasoning to find the Tempearture at the points in the cycle.
If actual numbers are given in the problem, calculate the numerical value of P, V, and T at the points on the cycle.
Make a First Law table.
Fill in zeroes during steps where there is no work - whenever $V$ is constant.
Calculate $\Delta E$ for each step by using the equipartition theorem $\Delta E = D\frac{1}{2}Nk_b \Delta T$ where D is the degrees of freedom for your substance - $D=3$ for a monatomic gas.
Use the First Law $\Delta E = Q + W$ to solve for the heat.
Recall that $ PV = nRT = Nk_B T$, so if you have the equipartition theorem that says $\frac{3}{2} Nk_B \Delta T = \Delta E$, you can relate that to $PV$, which makes adding up totals easier. Do not report Q totals as ex $3NRT - PV$, convert to the same state variables.
Fill in remaining spots on table and sum up totals.
*GOOD way to check your work: Make sure that total $\Delta E=0$.
Misconceptions & Mistakes
Pro Tips
Multiple Representations
Multiple Representations is the concept that a physical phenomena can be expressed in different ways.
Physical
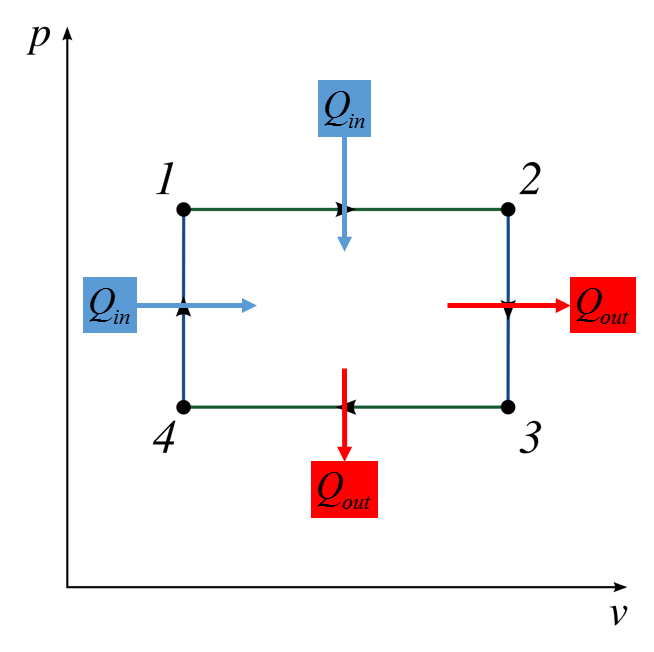
The diagrams below describe the heat engine process.The heat engine, or system takes in energy from a hot source where the heat energy is extracted to do work and waste leaves the system as used energy to the cold resevoir.
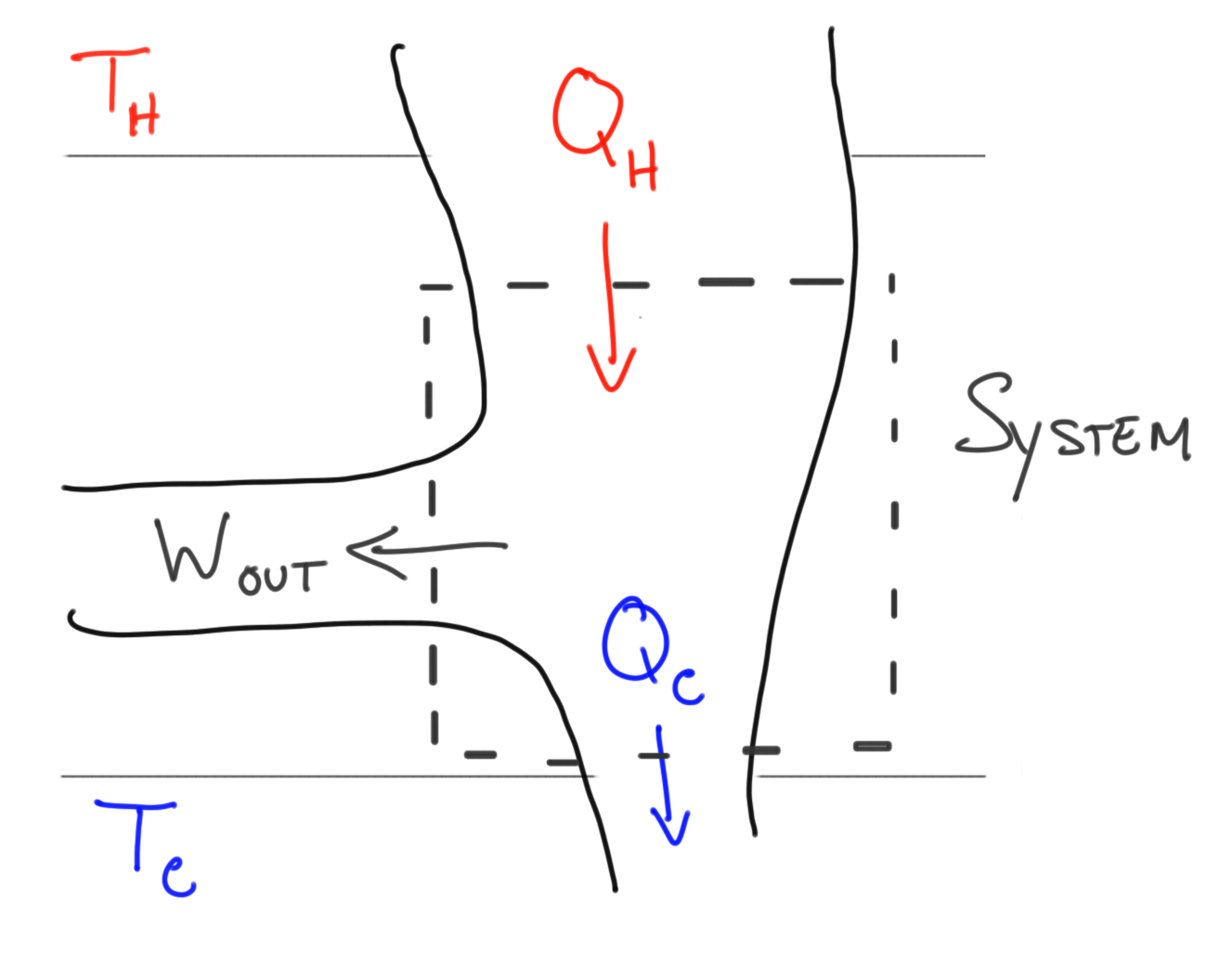
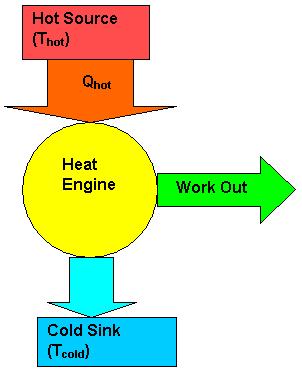
Mathematical
$\Delta E = Q+W$
$PV=NK_{b}T$
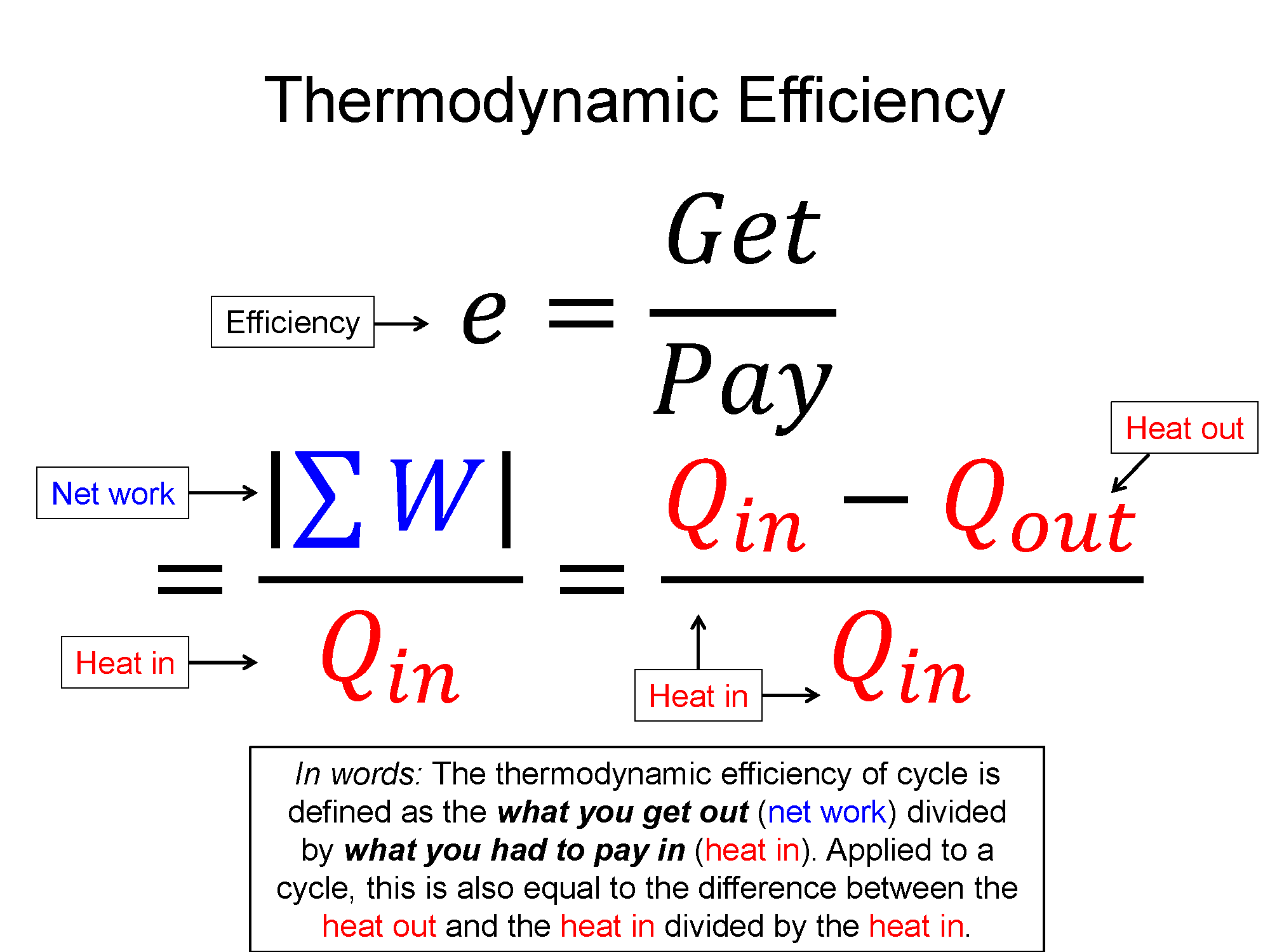
Efficiency: $ e = \frac{|\sum W|}{\sum Q_{in}}$
Isobaric
$\Delta P = 0$, then $\Delta V \propto \Delta t$
$W = P \Delta V$
Isothermal
$\Delta T = 0$, then $P \propto \frac{1}{V}$
$W = \frac{3}{2}nT ln(\frac{V_{f}}{V_{i}}))$
Adiabatic
$Q=0$, then $\Delta E = W$
Isochoric
$\Delta V = 0$ and $W=0$
Totale energy ($E$), Heat ($Q$), Work ($W$), Efficiency ($e$), Pressure ($P$), Volume ($V$), Time ($t$), Temperature ($T$),
Number of moles ($n$)
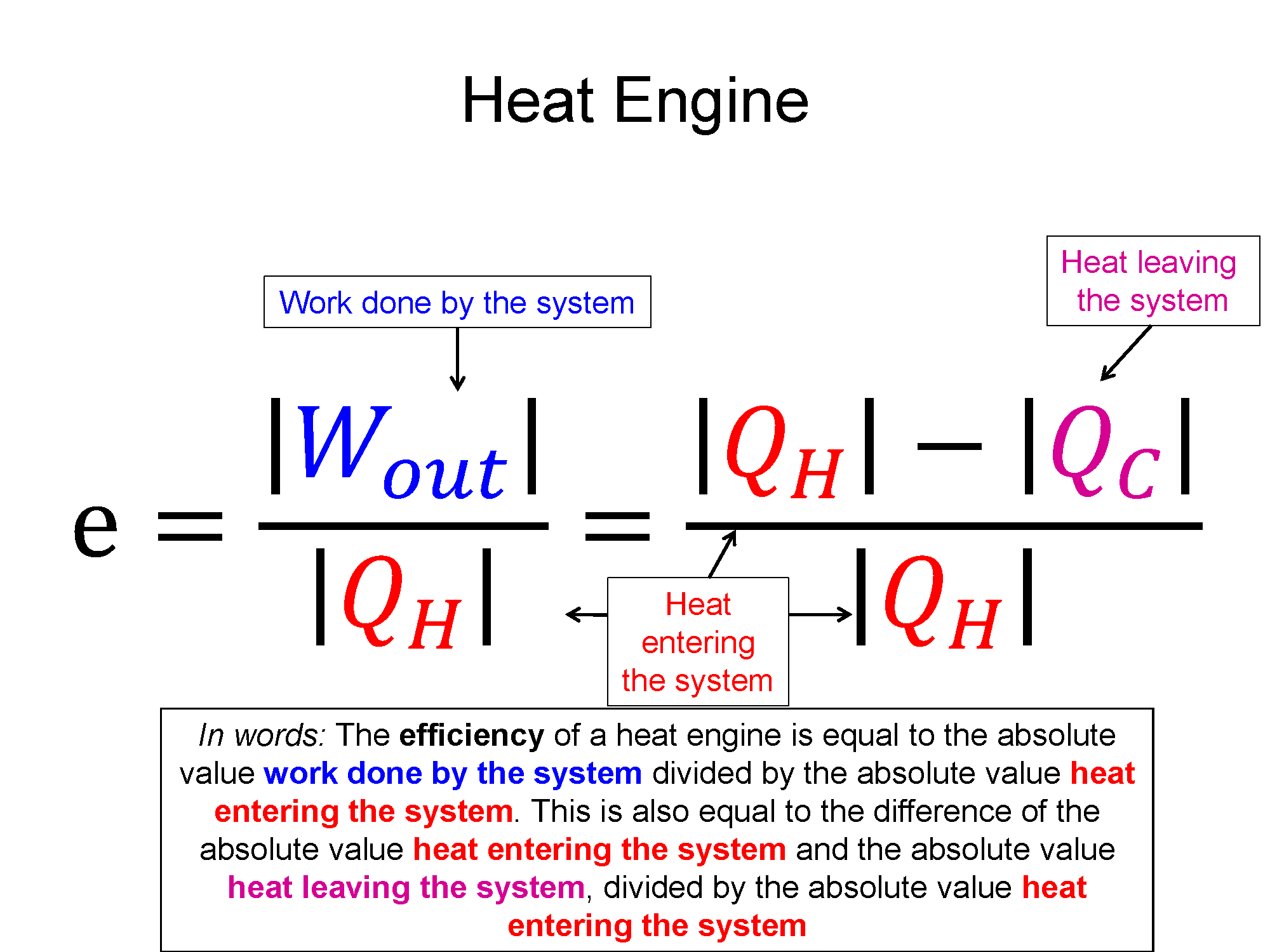
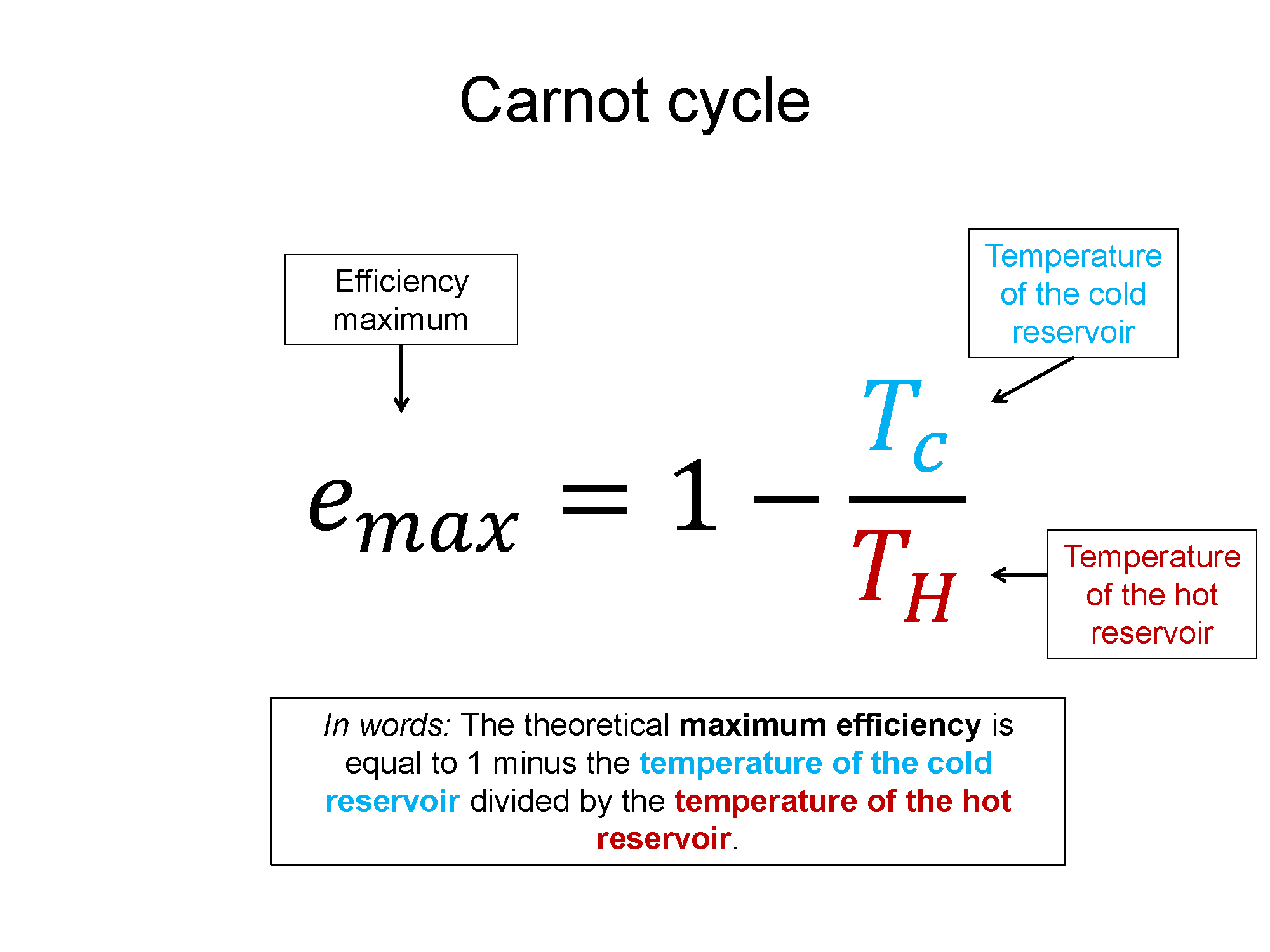
Graphical
1-2 Isobaric, 2-3 Isothermal
Descriptive
Here is a link to a description of the Carnot Cycle from Hyper Physics. This is an ideal description of a perfectly efficient cycle.
Experimental
The Experimental Representation for this would be to build a Stirling engine which is an application of the Carnot cycle.