Charges & the Electric Force: Fundamentals
Charges & the Electric Force: Fundamentals
BoxSand's Resources
Introduction
All of the forces we experience on a daily basis, except gravity, are due to the electric force. The electric force is a fundamental feature of charged particles. Everyday matter is largely comprised of these particles - the positively charged proton and negatively charged electron. If you're sitting in a chair, the force preventing you from falling towards the center of the Earth is due to repulsive force of the electrons on your bottom with the electrons on your seat. The electrons can't part ways and slide past each other, which would cause you to fall to the floor, because of their attraction to the protons in the nucleus. This balance of repulsion and attraction, is what causes all of the contact forces we experience (normal, friction, tension, buoyancy, etc.).
To understand how this works, we should start with some fundamental facts.
- Particles are either positive (protons), negative (electrons), or neutral (neutron).
- Charges are quantized - the charge of a proton or electron is $e=\pm 1.602\times 10^{-19}$ coulombs.
- Total net charge of a system is, $Q_{net}=e(N_{protons}-N_{electrons})$, where N represents the number of protons or electrons.
- Like charges repel each other while unlike charges attract each other.
- Charges are transferable from one object to another.
- Insulators are material with no free electrons and thus charges cannot flow freely throughout the material while a Conductor is a material with free electrons (unbound) that can move freely, spreading out uniformly throughout the material's surface.
Conductors vs. Insulators
Most electrons are bound to a particular atom but their are some electrons that will be transferred to another atom or an entirely different object. Understanding how charge transfer works starts by understanding the two main types of materials, insulators and conductors. Both materials have fairly fixed positive ion cores (protons in the nucleus) that are much heavier (x1000) than the electrons surrounding them. Both have tightly bound electrons in the inner orbital shells.

Where they differ is in the presence of free electrons. Conductors have electrons that are bound enough to stay on the material but not bound too strongly to any one atom. They can move freely, allowing them to redistribute throughout an object quickly if initially concentrated in one location. Metals are often conductors and are used in electric wiring. Insulators on the other hand have no free electrons and as such do not allow current to flow through them easily. The rubbery material surrounding the electrical wires are insulators to prevent you from getting shocked.
Charge Transfer
Electrons can be transferred between objects allowing for them to be in a net negative or net positive state. If two objects are initially net neutral, but after charge transfer occurs between them one is net positive, you know the other must be equally net negative. This is because one loses electrons and the other gains electrons - the protons in the nucleus are not directly involved in any charge transfer.
Conductors can exchange charges by simply touching due to their free electrons. If touched, both objects will come to an equivalent equilibrium charge distribution. Consider two metal spheres, equivalent in every way except their charge, being brought into contact. If one was initially charged with a net charge $Q_{net}=+4 C$ and the other $Q_{net}=-2 C$, they have a net initial charge of $+2 C$. Since they have the same surface area, they will split the net charge equally, $+1 C$ each. The change will happen very rapidly, with the charges transferring between objects and redistributing facilitated by the free electrons in the conductors.
Insulators can have some charge transfer by simple contact but friction is often required for any large effects. Take a plastic rod and some cat fur and rub them together and one will lose electrons while the other gains them. Which one gains or loses an electron depends on the relative electronegativity of each material. Insulators differ from conductors in that the exchange of electrons only effects the region where the touching occurred. Since there are no free electrons to redistribute the excess charges, only the top half of the plastic rod would display charged effects if that was the only half rubbed.
Electric Force
To understand the effects of objects with a distribution of charges exerting a force on each other, lets start with the force between two point charges. This is known as Coulomb's Law. The below equation reads, the magnitude of the electric force of particle 2 on 1 is equal to the magnitude of the electric force of 1 on 2 (Newton's 3rd law). That force is equal to the electric constant $k$ times the magnitude of $q_1$, times the magnitude of $q_2$, divided by the absolute distance between $q_1$ and $q_2$ squared.
$|\overrightarrow{F}^E_{21}| = |\overrightarrow{F}^E_{12}| = \frac{k|q_1||q_2|}{|\Delta \overrightarrow{r}_{12}^2|}$, where $k=8.99\times 10^9 N \cdot m^2/C^2$
The direction of the net force is determined by drawing a Free-Body Diagram and using the fact that unlike charges attract and like charges repel.
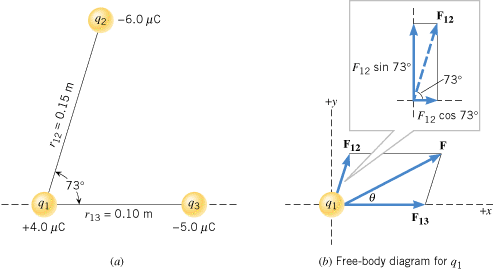
Here you see an example of the FBD for $q_1$ interacting with $q_2$ and $q_3$. Since 1 is positively charged and 2 and 3 are both negatively charged, 1 is attracted to both 2 and 3. So both $F_{12}$ and $F_{13}$, acting on particle 1, are towards the other particle. The magnitude of $F_{13}$ is larger than $F_{12}$ even though $q_2$ has a greater charge. That is due to $q_3$ being closer to $q_1$ than $q_2$. With the electric force being inversely proportional to the distance between the particles and only proportional to the charge to the 1st power, being closer means a greater force in this case.
To find the force on a charged particle you typically have to do the following:
- Draw a FBD of forces acting on the particle of interest using whether they attract or repel and a rough scaling of how large due to the relative charge magnitudes and distances.
- Determine the distance between the particle of interest and each other particle.
- Determine the magnitude of each electric force.
- Use vector summation to add up the components of each electric force and find the net force. You may have to use an angle found using geometry of the physical setup to find a component of the force.
Now a balloon that's charged and is attracted to a wall is not point charges but is comprised of point charges. The mathematics can get more tedious for a collection of charges distributed around an object but the fundamental principles hold true. For a more sophisticated and powerful analysis of the electric force, the concept of the electric field was developed. Read about the connection between the field and the force in the section on Electric Fields.

Videos
Pre-lecture Videos
Watch these videos before doing the pre-lecture assignment, ** denotes supplemental but suggested
Introduction to E-force, q-transfer, insulators vs conductors(50min)
electroscope, polarization(22min)
Coulombs law with example (12min)
Web Resources
Text
Openstax has three sections covering Electricity, Charge, and Coulombs Law.
|
Static Electricity and Charge |
Conductors and Insulators | Coulomb's Law |
Boston University's Page on electric charge and Coulomb's law is neat reference with a couple of example problems
PPLATO is a complete resource with a lot of information, and several practice questions per subject. This webpage covers electric charge, the electric field, and electric potential, frocus on the electric charge, we'll get back around to field and potential later.
The Physics Classroom has an entire chapter on static electricity. The last lesson of the chapter is on Electric Fields so save that for later!
Other Resources
This link will take you to the repository of other content related resources .
Videos
Pre-Med Academy is back with three well put together videos on our main subjects, Electric Charge, Electric Force, and Coulomb's Law
Step By Step Science has several example problems, here are a few on using Coulomb's Law. There are more in this section's content repository.
Other Resources
This link will take you to the repository of other content related resources .
Simulations
This simulation by Duffy helps you with Coulombs Law and Newton's 2nd law
The colorado PhET simulation is everything you ever needed in life, that you didn't know that you needed. We present to you, John Travoltage.
Charge transfer PhET simulation with baloons and a sweater.
For additional simulations on this subject, visit the simulations repository.
Demos
Rub a plastic cup against carpet, hair, or fur, and observe the effects of the net charge now on the cup.

For additional demos involving this subject, visit the demo repository
Practice
Fundamental examples
(1) A point charge with charge $q_1 = 1.8 nC$ is located at the origin. A point charge with charge $q_2 = 3.6 nC$ is located a distance $r_{12} = 6 nm$ away. (a) What is the magnitude of the coulomb force from charge 1 on charge 2? (b) Is this an attractive or repulsive force?
(2) A point charge with charge $q_1 = - 1.8 nC$ is located at the origin. A point charge with charge $q_2 = 3.6 nC$ is located a distance $r_{12} = 6 nm$ away. What is the magnitude of the coulomb force from charge 1 on charge 2?
(3) A point charge with charge $q_1 = - 1.8 nC$ is located at the origin. A point charge with charge $q_2 = 3.6 nC$ is located a distance $r_{12} = 6 nm$ away. A point charge with charge $q_3 = 5 nC$ is located at a position $r_3 = <2,2> nm$. What is the magnitude of the coulomb force from charge 1 on charge 2?
Solutions found HERE
Short foundation building questions, often used as clicker questions, can be found in the clicker questions repository for this subject.
Practice Problems
BoxSand practice problems - Answers
BoxSand's multiple select problems
BoxSand's quantitative problems
Recommended example practice problems
- OpenStax, Period and Frequency also linked in Charges & the Electric Force sections.
- Static Electricity and Charge, Website Link
- Conductors and Insulators, Website Link
- Coulomb's Law, Website Link
- Large set of questions on electric charge and force. External PDF
- Coulomb's Law Review, External PDF
For additional practice problems and worked examples, visit the link below. If you've found example problems that you've used please help us out and submit them to the student contributed content section.